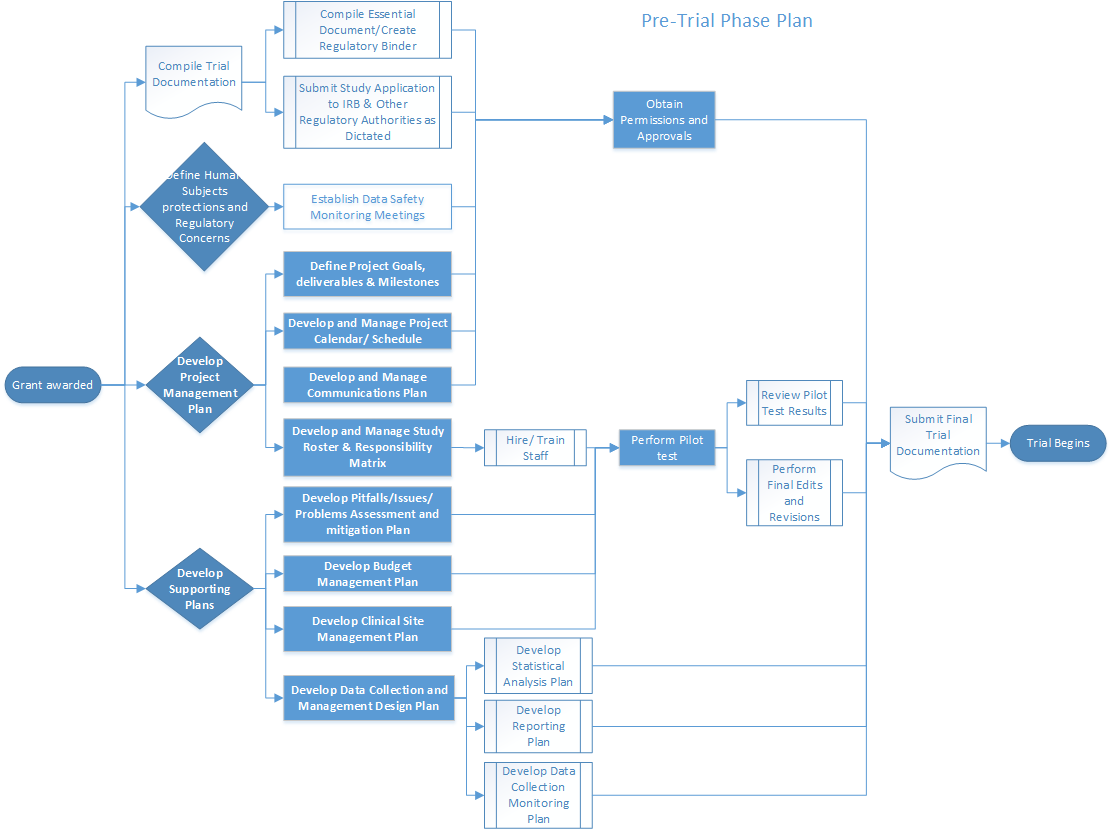
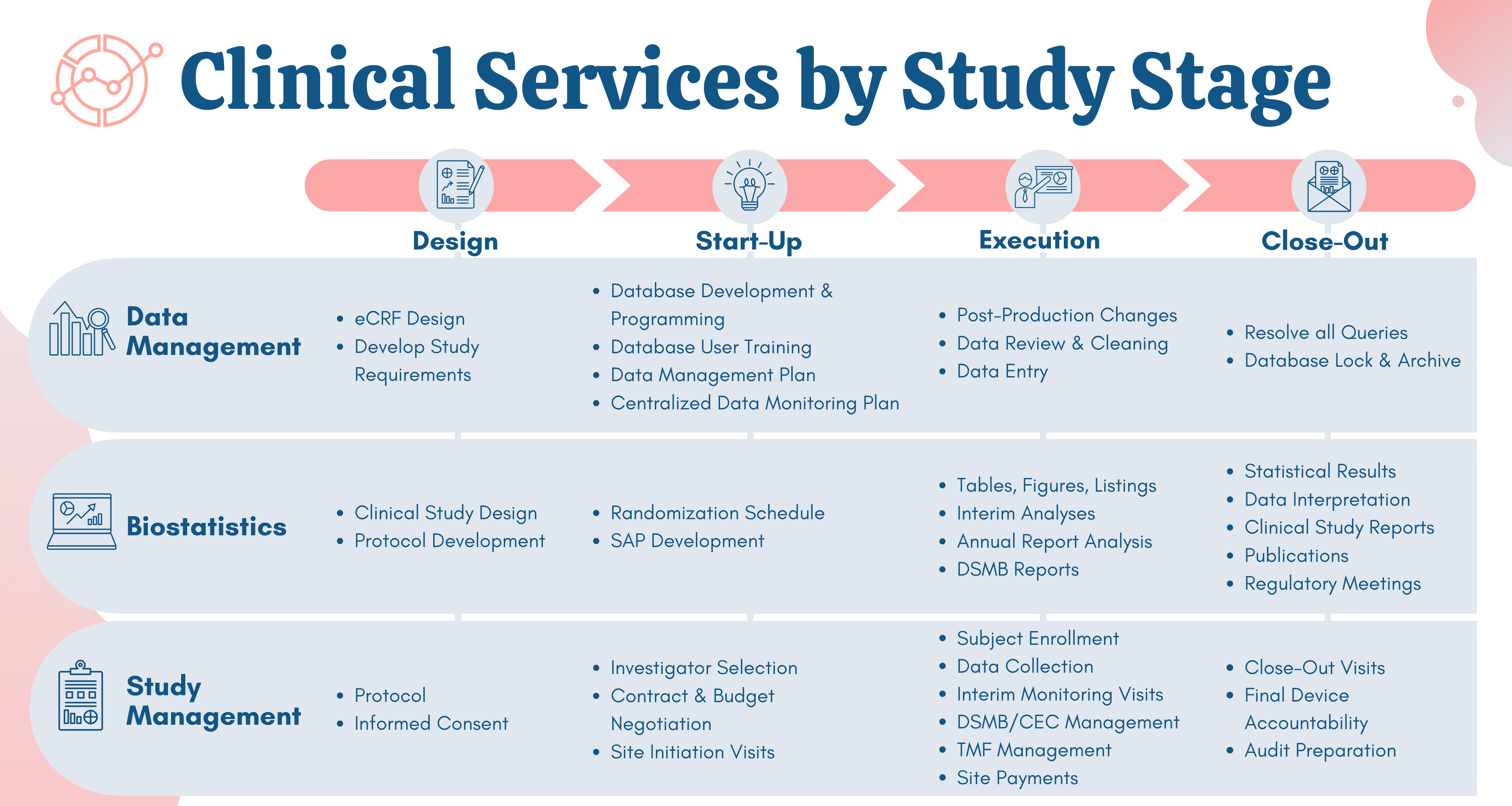
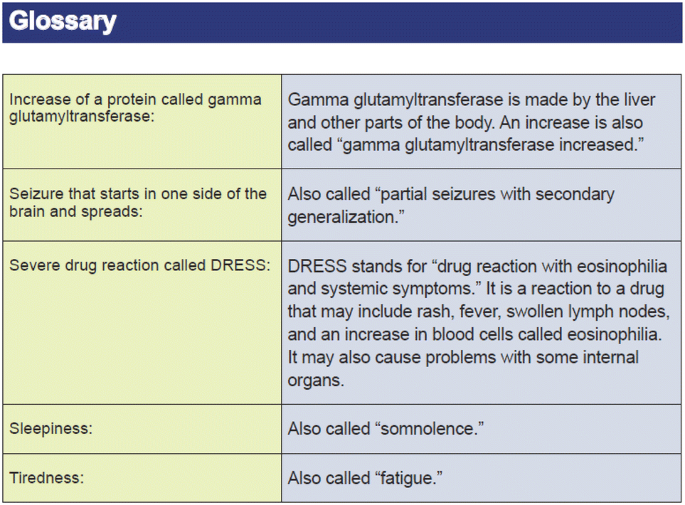
Completeness of Reporting of Patient-Relevant Clinical Trial Outcomes: Comparison of Unpublished Clinical Study Reports with Publicly Available Data | PLOS Medicine

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet
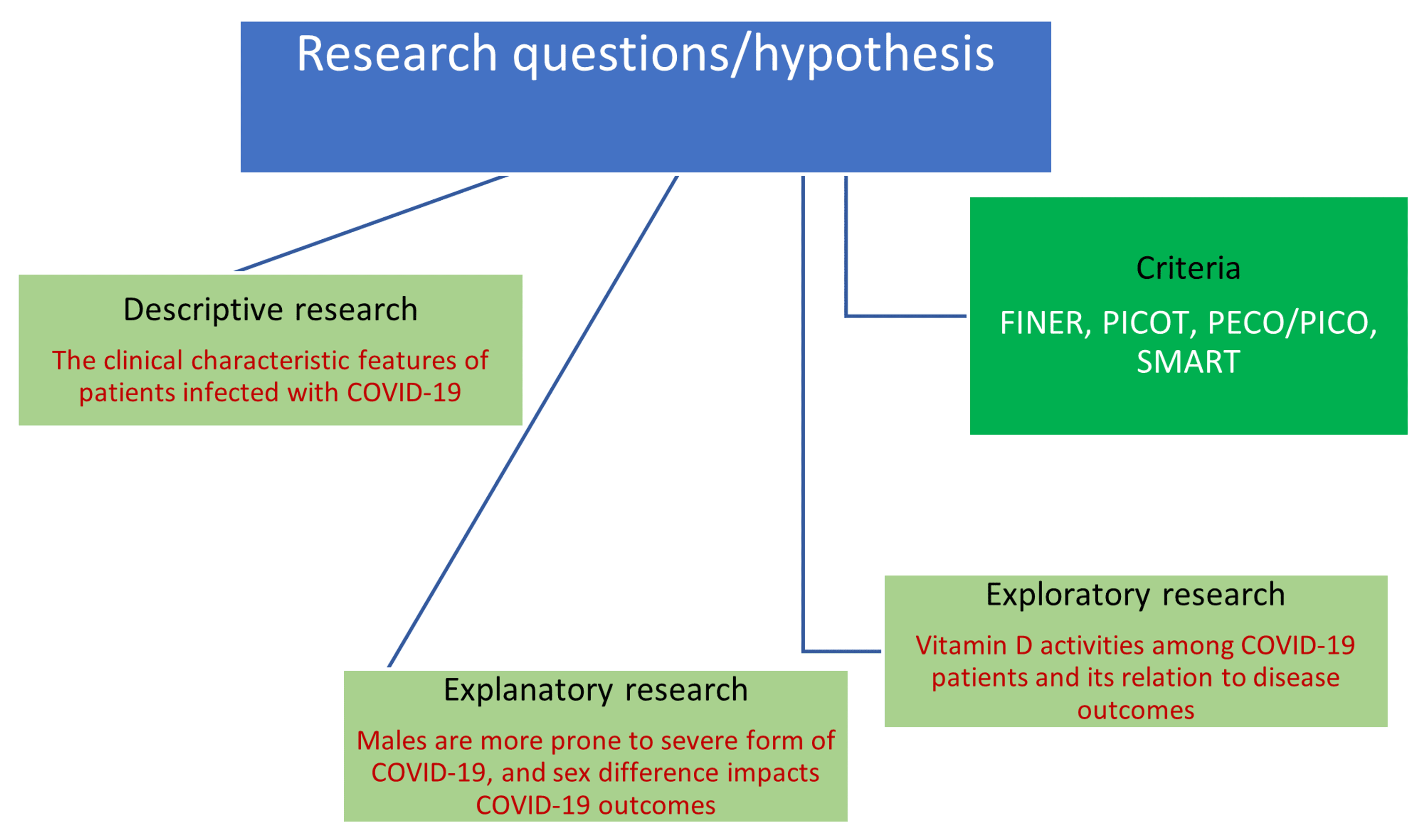
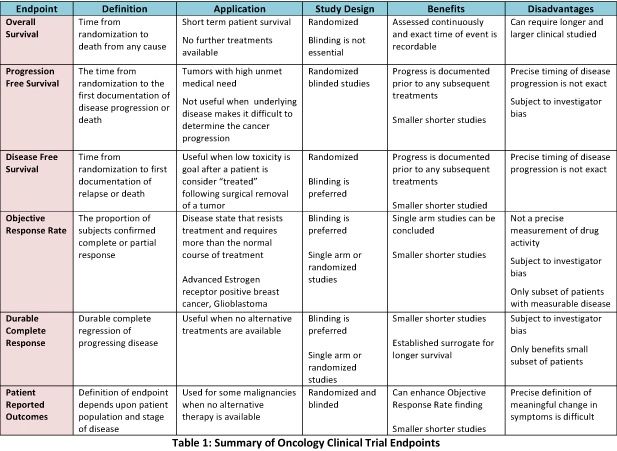
Cureus | Research Question, Objectives, and Endpoints in Clinical and Oncological Research: A Comprehensive Review | Article