Aβ Oligomers Cause Localized Ca2+ Elevation, Missorting of Endogenous Tau into Dendrites, Tau Phosphorylation, and Destruction of Microtubules and Spines | Journal of Neuroscience

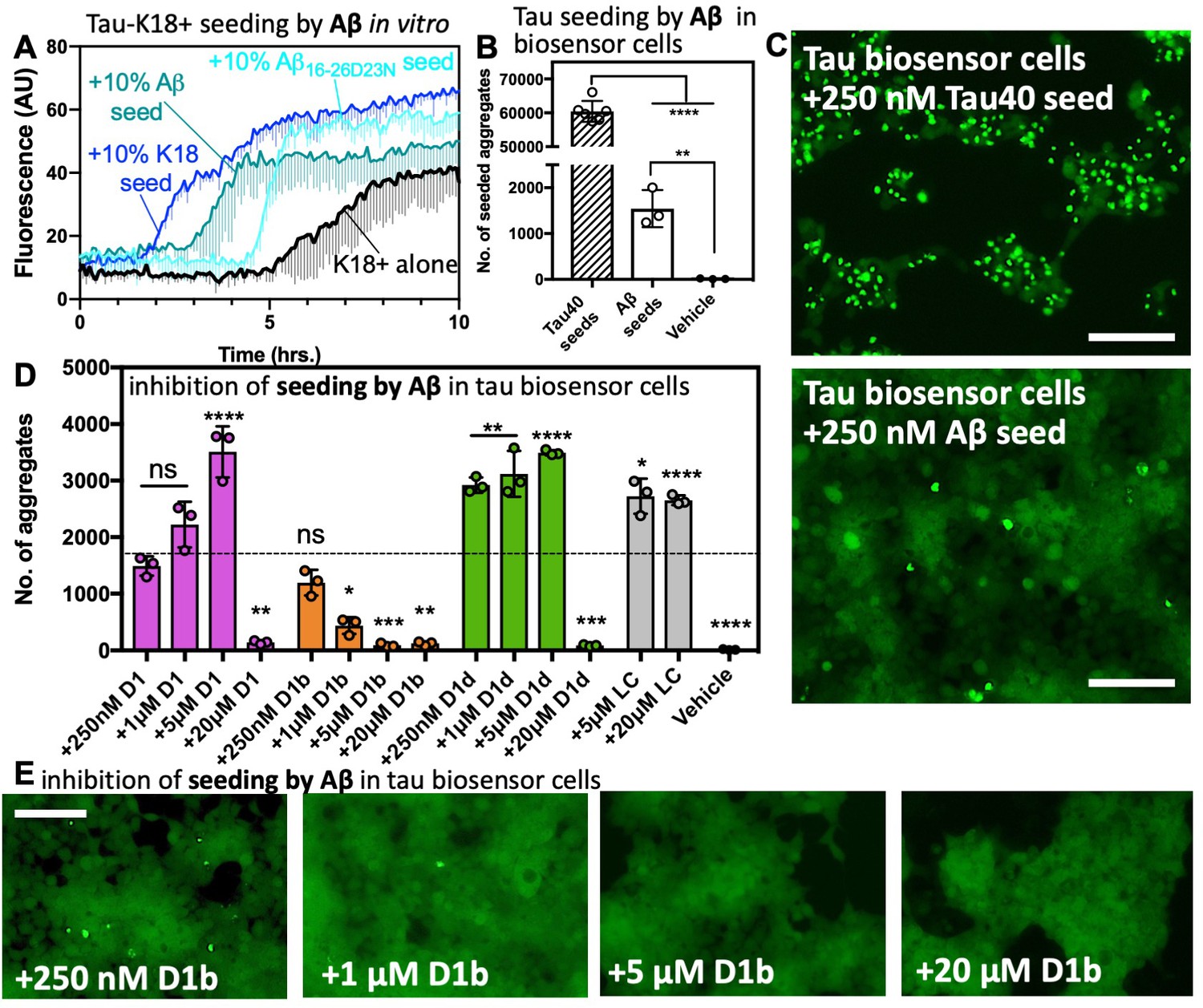
Aβ Oligomers Cause Localized Ca2+ Elevation, Missorting of Endogenous Tau into Dendrites, Tau Phosphorylation, and Destruction of Microtubules and Spines | Journal of Neuroscience
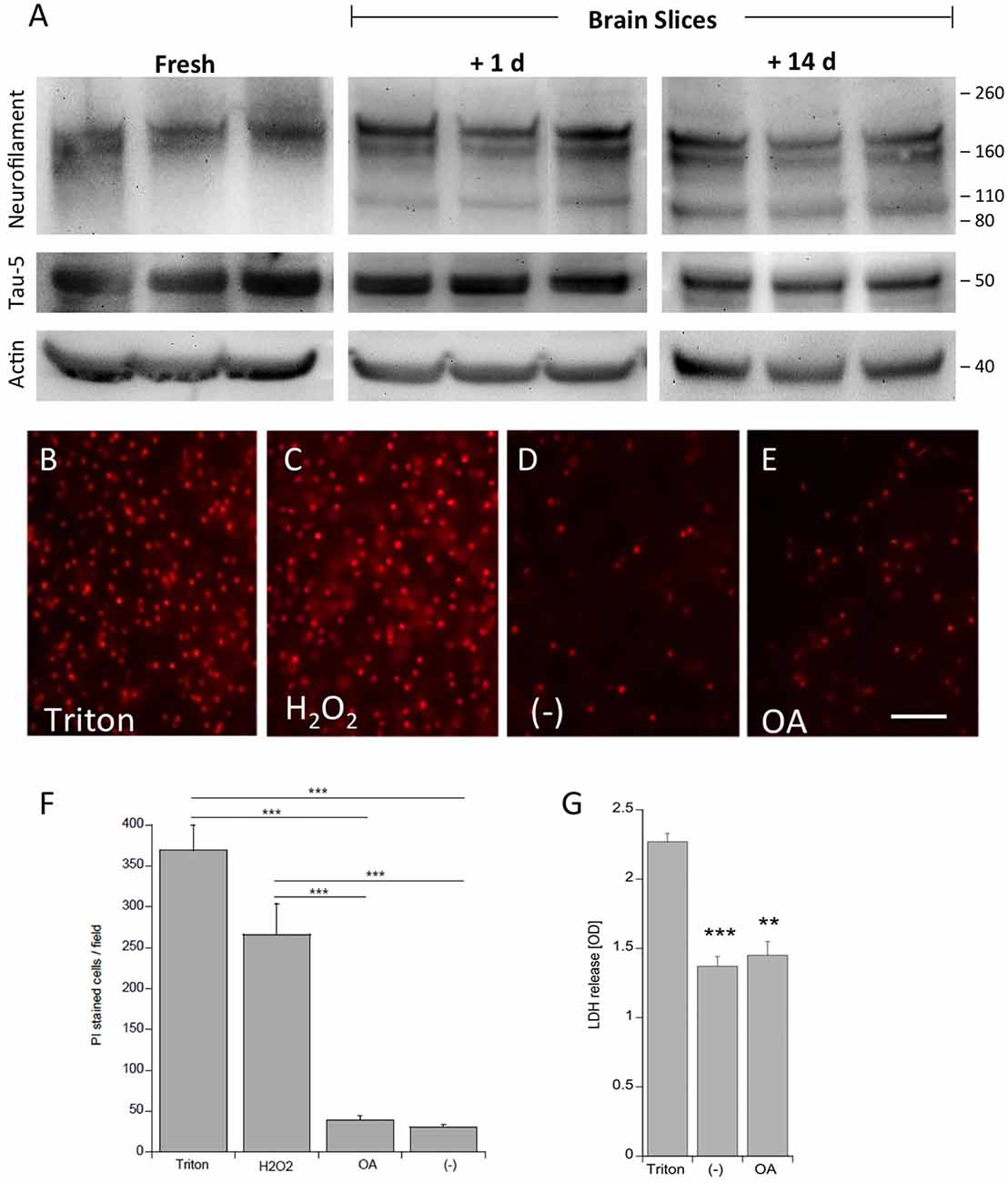
Frontiers | Differential Hyperphosphorylation of Tau-S199, -T231 and -S396 in Organotypic Brain Slices of Alzheimer Mice. A Model to Study Early Tau Hyperphosphorylation Using Okadaic Acid

Simple, Reliable Protocol for High-Yield Solubilization of Seedless Amyloid-β Monomer | ACS Chemical Neuroscience

Rapid Tyrosine Phosphorylation of Neuronal Proteins Including Tau and Focal Adhesion Kinase in Response to Amyloid-β Peptide Exposure: Involvement of Src Family Protein Kinases | Journal of Neuroscience

Dendritic Function of Tau Mediates Amyloid-β Toxicity in Alzheimer's Disease Mouse Models - ScienceDirect

C-Terminally Truncated Forms of Tau, But Not Full-Length Tau or Its C-Terminal Fragments, Are Released from Neurons Independently of Cell Death | Journal of Neuroscience

Dendritic Function of Tau Mediates Amyloid-β Toxicity in Alzheimer's Disease Mouse Models - ScienceDirect

Aβ Oligomers Cause Localized Ca2+ Elevation, Missorting of Endogenous Tau into Dendrites, Tau Phosphorylation, and Destruction of Microtubules and Spines | Journal of Neuroscience

Motorcycle Rear Wheel Hub Rubber Buffer for Honda C70 Passport S90 CL90 CT90 S65 Rear Sprocket Damper Cushion - AliExpress
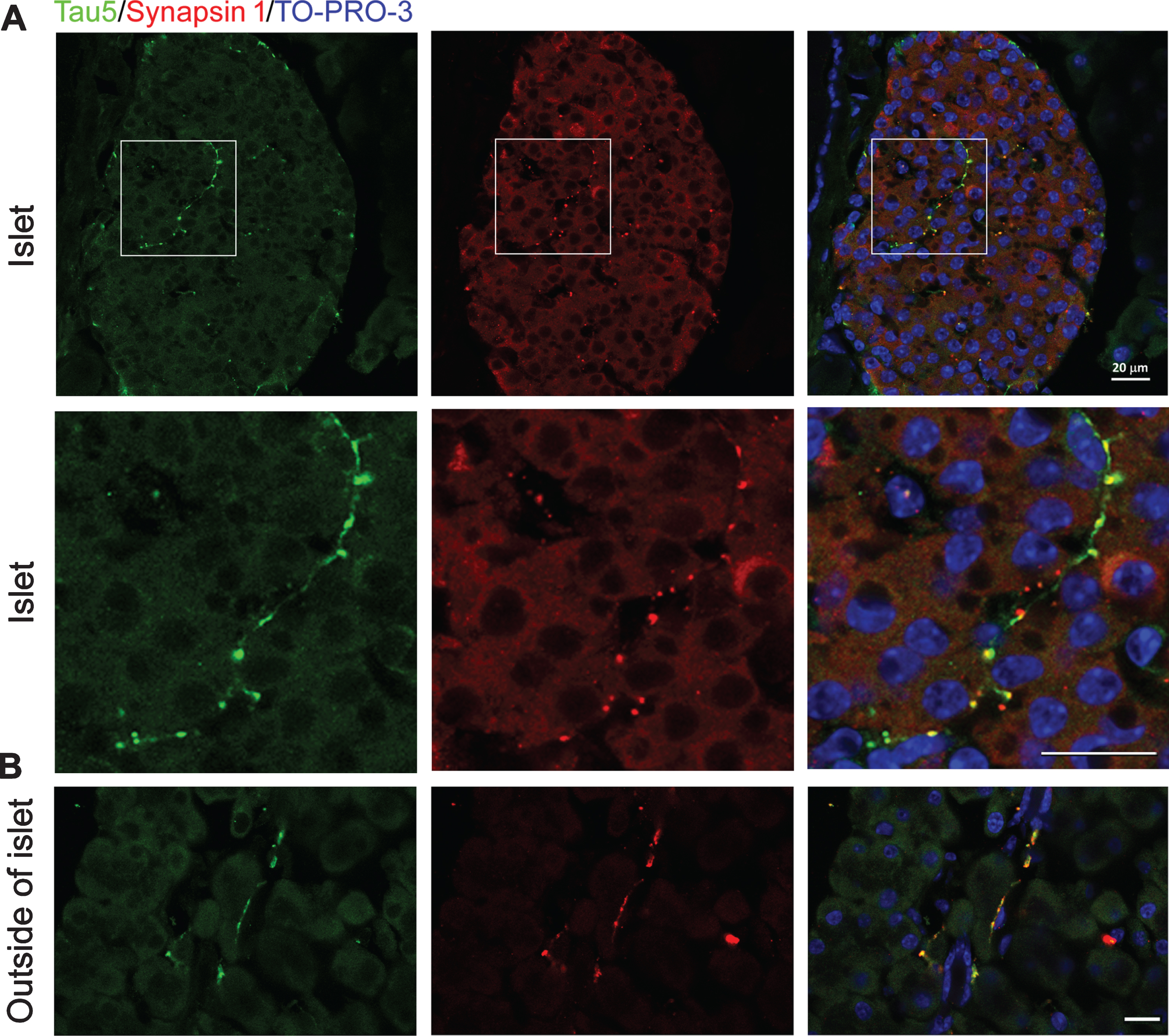
Expression of Microtubule Associated Protein Tau in Mouse Pancreatic Islets Is Restricted to Autonomic Nerve Fibers - IOS Press